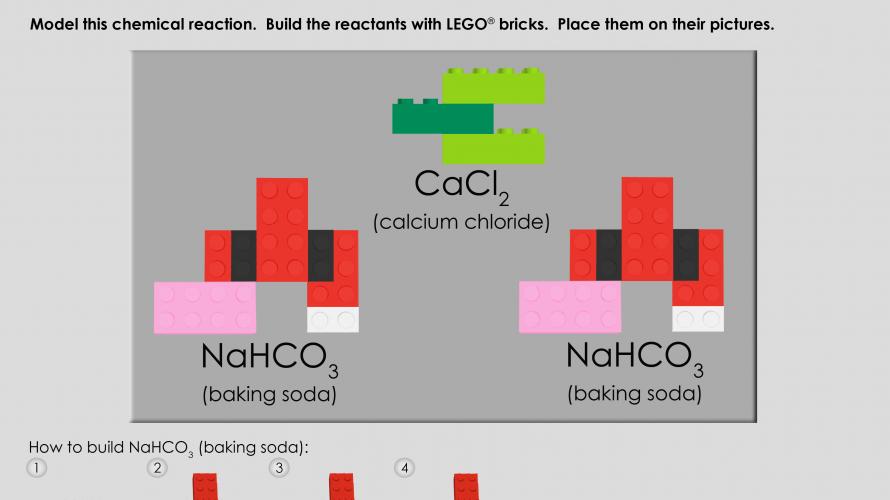
Students are introduced to atoms, molecules, compounds, and mixtures, using LEGO bricks as atoms. In an engaging hands-on wet lab, students experience a chemical reaction and then model the same reaction with LEGO bricks.
Chemical Reactions
This unit is appropriate for children ages 11 and up. Students are introduced to atoms, molecules, compounds, and mixtures, using LEGO® bricks as atoms. In an engaging hands-on wet lab, students experience a chemical reaction and then model the same reaction with LEGO bricks. This unit is also offered as a 3-hour academic field trip LEGO Chemistry at the Edgerton Center.
For more information:
Molecule Sets:
You can make your own Molecule Sets by visiting Information for Edgerton Center Molecule Sets. We are no longer able to sell Molecule Sets. We recommend students work in pairs with 1 kit.
The following LEGO bricks are the minimum required for the Chemical Reactions Lesson:
- 6 red 2x4
- 2 pink 2x4
- 2 light green 2x4
- 2 black 2x4
- 1 green 2x4
- 4 white 1x2
NOTE: "2x4" and "1x2" refer to the number of bumps on top of the LEGO bricks.
Chemical Reactions Mats:
Print all mats in color at 100% size on 8.5"x11" paper. The Atom Key/Layout Mat is required for all Molecule Set lessons.
Student Worksheets:
- Chemical Reactions Student Pages (Color)
- Chemical Reactions Student Pages (B&W)
- Chemical Reactions Student Key
Wet Lab Materials:
The required materials for the wet lab portion of this lesson are in the Chemical Reactions Teacher Guide (link)
Substitute Chemical Reactions:
Instead of using baking soda and calcium chloride, this lesson can be taught using baking soda and vinegar as the reactants instead. This reaction is simpler and requires fewer wet lab materials and different LEGO bricks. See Baking Soda and Vinegar Mat
Phenol red can also be substituted with red cabbage juice.
Co-Teaching Video from BLOSSOMS:
This 25-minute, interactive video can be used to co-teach the lesson with Dr. Kathleen Vandiver (inventor of this lesson). It includes a teacher guide, transcript, and additional resources. Alternatively, teachers can watch the entirety of the lesson in advance. BLOSSOMS (Blended Learning Open Source Science or Math Studies) is a collaborative initiative seeking to begin to develop a large, free repository of video modules for high school math and science classes in multiple languages.
Chemical Reactions Teacher Guide:
This teacher guide outlines the lessons as multiple 45-minute class sessions for both the wet lab and LEGO activity. Alternatively, the guide contains a workshop (2.5 hour) version.
MIT Edgerton Center videos on our YouTube channel:
This 7.5-min video was designed as a preview for instructors before teaching the wet lab. It shows all the experiments in the wet lab and the expected results. If necessary, it might be used to replace the wet lab activity.
Massachusetts State Frameworks for grades 6-8, Physical Sciences Strand: Elements, Compounds & Mixtures: (review - find energy standards)
5. Recognize that there are more than 100 elements that combine in a multitude of ways to produce compounds that make up all of the living and nonliving things that we encounter.
6. Differentiate between an atom (the smallest unit of an element that maintains the characteristics of that element) and a molecule (the smallest unit of a compound that maintains the characteristics of that compound).
7. Give basic examples of elements and compounds.
8. Differentiate between mixtures and pure substances.
10. Differentiate between physical changes and chemical changes.
Next Generation Science Standards (NGSS):
MS-PS1.B.1: Substances react chemically in characteristic ways. In a chemical process, the atoms that make up the original substances are regrouped into different molecules, and these new substances have different properties from those of the reactants.